|
According to the CS2 Lewis structure, carbon is the central atom with zero lone pair of electrons and two sulphur atoms attached to it. HybridizationĪccording to the VSEPR theory, the space number is used to determine the hybridisation of the atom. Therefore, there are two regions of electron density around the carbon centre atom, which, according to the VSEPR theory, will give a linear molecular geometry. This is because the carbon (C) centre atom has no lone pair of electrons and is attached to two sulphur (S) atoms. Both atoms (carbon and sulphur) have completed their octet rules and both have reached a stable structure. After the move, each of the two sulphur atoms forms a double bond with the carbon atom, retaining two lone pair electrons on each sulphur atom. In order to stabilise this carbon atom, the electron pair has to be moved away from the outer sulphur atom so that the carbon atom can have eight electrons (i.e. Therefore, to overcome this problem, we will take the help of sulphur lone pair of electrons. 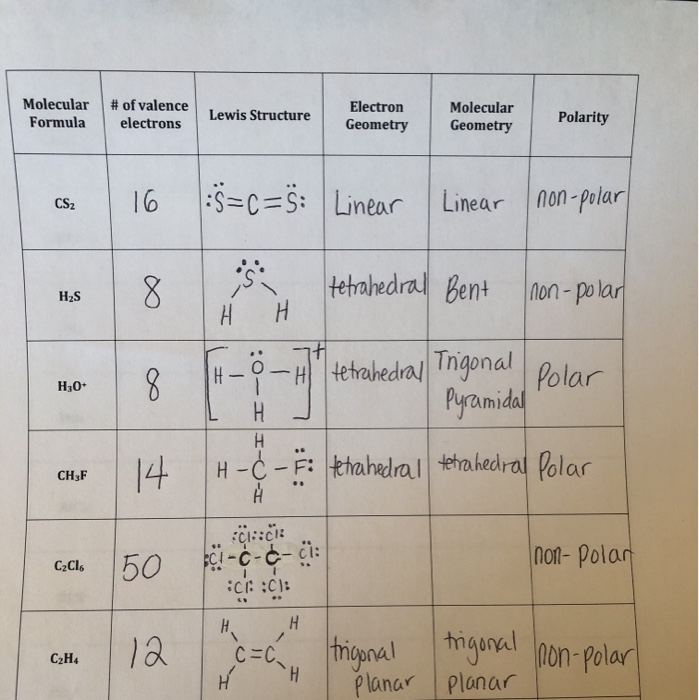
In order to check the stability of the central carbon (C) atom, we have to check whether it is forming an octet or not, moreover, we do not have extra valence electrons for completing the octet of carbon. Therefore, We should try to reduce charges on atoms if it is a possible. When there are positive and negative charges on lot of atoms or higher charges (like +2, +3, -2, -3) on atoms in an ion or molecule, that structure is not stable. Step 4 Stability of structure and minimize charges on atoms by converting lone pairs to bonds Thus, there are 3 lone pairs of electrons on each sulfur atom (2). 
The carbon atom is connected to each of the 2 sulphur atoms by a σ-bond (one σ-bond equals one pair of electrons), and since we are left with 12 valence electrons, we must first place these electrons around the outer atom (sulphur) in order to complete its octet rule. For the CS2 molecule, the total number of pairs of electrons is eight. Total number of valence electron pairs = σ-bonds + π-bonds + lone pairs of electrons in the valence layer, i.e., the total number of valence electron pairs divided by 2. Step 3 Labelling the electron lone pairs between atoms For the CS2 molecule, the carbon atom (C) is less electronegative than the sulphur atom, so carbon is the central atom and sulphur is the outer atom. The central atom must be highly or minimally electronegative. Therefore, there are 4 and 6 valence electrons in the carbon and sulphur atoms respectively, so the total number of valence electrons in the CS2 molecule = valence electrons from 1 carbon atom + valence electrons from 2 sulphur atoms = 4 + 6(2) = 16. Step 1 Calculate the number of valence electrons for C and SĬarbon and sulphur are elements of groups 14 and 16 of the periodic table. Steps for drawing the CS2 Lewis structure Each sulphur atom (S) has two lone pairs of electrons. The carbon (C) and sulphur (S) atoms are each connected by a double bond. 
The CS2 Lewis structure consists of a central carbon atom (C) and two external sulphur atoms (S) bonded at 180°.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
