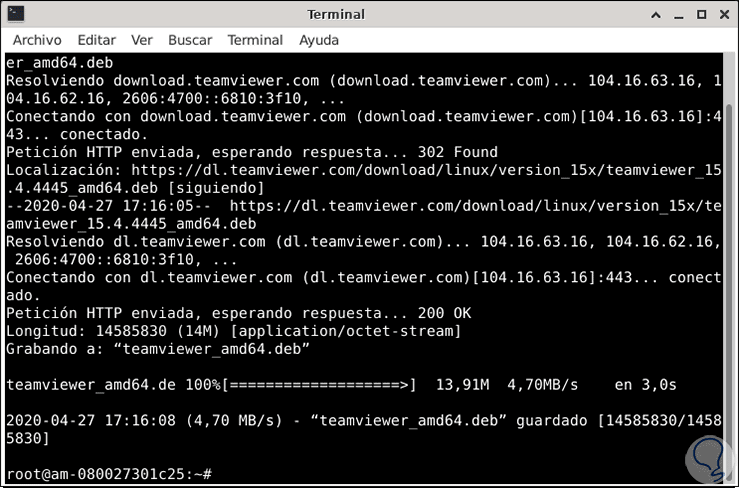
We have successfully installed Teamviewer on Ubuntu. They’ll be asked for a password later, after which they’ll be able to access your PC remotely.Īnd that’s it. The user will enter the ID in the text area labeled “ Insert partner ID,” then click the “ Connect” button. To connect to a remote user, provide your TeamViewer ID and password. Once you launch the application, agree to the EULA agreement.įinally, you will be able to access TeamViewer’s user interface. $ teamviewerĪlternatively, you can search for and launch the TeamViewer application via the application manager, as shown below: To launch TeamViewer, run the following command on your terminal. Now that the installation is done, TeamViewer is now ready to use. teamviewer_bĭependency packages that are required by TeamViewer will be downloaded during the installation process. To do so, run the following apt command on the Debian package that you downloaded moments ago. Now we can proceed to install TeamViewer on Ubuntu Linux. Step 3) Install TeamViewer with apt command Run the below wget command to download TeamViewer’s Debian package: $ wget Next, we download the latest version of TeamViewerfrom the official site. $ sudo reboot Step 2) Download TeamViewer on Ubuntu Linux Reboot the system once all the updates are applied successfully. To update your system run: $ sudo apt update It is an essential tool for sharing files or fixing each other’s computing machine’s problems through remote connections.It is always important to ensure your system is updated before proceeding with a new installation. TeamViewer can be installed on Debian using its “.deb” package file from its official website. Moreover, if you want to get TeamViewer on Ubuntu (the most used Debian-based distribution), read our article. Note: All the methods and steps illustrated in this post are practically performed on the latest release of Debian, i.e., Debian 11. The autoremove will remove all the relevant data of the TeamViewer application from Debian. If you are notified of installation failure due to missing dependencies, type the below command to complete the installation. If you are no more using TeamViewer on your system, then it is recommended to remove it by following the command below. Alternatively, if you prefer the terminal way, navigate to the download folder and run the below command: sudo dpkg -i teamviewer. The users can share their remote-id and password to allow the other TeamViewer user to connect with them. $ teamviewerĪfter accepting the license terms and conditions, the TeamViewer was launched, and the interface can be seen in the following image. Once the installation is complete, you can start TeamViewer by typing the following command in the terminal. $ sudo apt -f install Launching TeamViewer If you see any errors or dependencies, run the following command to fix them. All Categories 26. Note: Replace “teamviewer_b” with the actual name of the DEB package you downloaded. In an other way can you tell me what packages and dependencies are needed to run TeamViewer on debian considering that almost no extra stuff are installed on our minimal OS. You can also use the “dpkg -i” command, which is old and has lesser functionality than “apt install”. Just run the command provided below to install TeamViewer. We have downloaded it through the terminal, so it has landed in our home directory. This article applies to TeamViewer (Classic) users on Linux.If you have downloaded the Debian package file through the GUI, it will be placed in your “Downloads” directory. 
Now, navigate to the directory where you downloaded the DEB package. Or you can download the command mentioned here directly from the terminal. Im trying to install teamview 12 in my debian 9 linux on an old laptop. Navigate to the download section on the official website of TeamViewer and download the Debian package. The TeamViewer will be installed using its “.deb” package support available on the official website. The Debian-based distributions support the “.deb” package support, and various applications/tools provide the installation package as a “.deb”. The steps provided in this post are practically demonstrated on Debian. This guide will provide a detailed installation procedure of TeamViewer for the Debian distribution. You can use it to provide technical support to someone else or to collaborate on projects with colleagues. TeamViewer is a remote-control software to access and control other computing machines remotely.
0 Comments
According to the CS2 Lewis structure, carbon is the central atom with zero lone pair of electrons and two sulphur atoms attached to it. HybridizationĪccording to the VSEPR theory, the space number is used to determine the hybridisation of the atom. Therefore, there are two regions of electron density around the carbon centre atom, which, according to the VSEPR theory, will give a linear molecular geometry. This is because the carbon (C) centre atom has no lone pair of electrons and is attached to two sulphur (S) atoms. Both atoms (carbon and sulphur) have completed their octet rules and both have reached a stable structure. After the move, each of the two sulphur atoms forms a double bond with the carbon atom, retaining two lone pair electrons on each sulphur atom. In order to stabilise this carbon atom, the electron pair has to be moved away from the outer sulphur atom so that the carbon atom can have eight electrons (i.e. Therefore, to overcome this problem, we will take the help of sulphur lone pair of electrons. 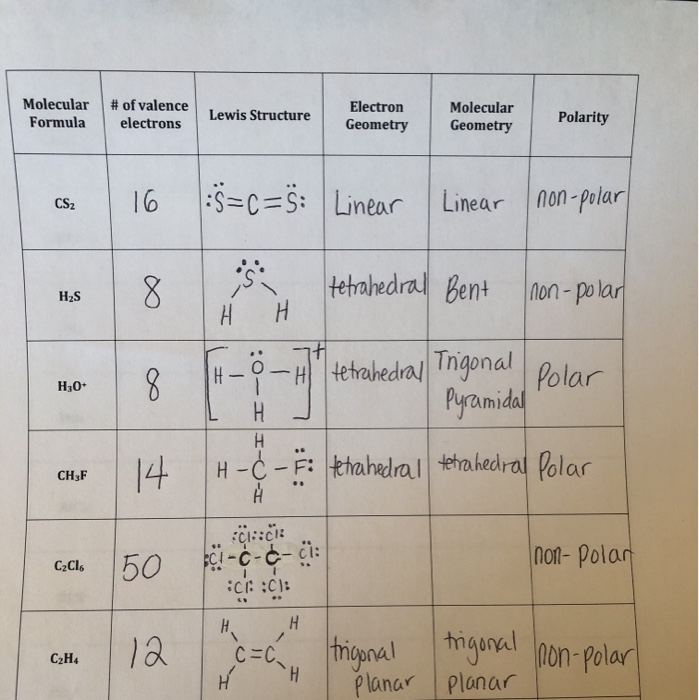
In order to check the stability of the central carbon (C) atom, we have to check whether it is forming an octet or not, moreover, we do not have extra valence electrons for completing the octet of carbon. Therefore, We should try to reduce charges on atoms if it is a possible. When there are positive and negative charges on lot of atoms or higher charges (like +2, +3, -2, -3) on atoms in an ion or molecule, that structure is not stable. Step 4 Stability of structure and minimize charges on atoms by converting lone pairs to bonds Thus, there are 3 lone pairs of electrons on each sulfur atom (2). 
The carbon atom is connected to each of the 2 sulphur atoms by a σ-bond (one σ-bond equals one pair of electrons), and since we are left with 12 valence electrons, we must first place these electrons around the outer atom (sulphur) in order to complete its octet rule. For the CS2 molecule, the total number of pairs of electrons is eight. Total number of valence electron pairs = σ-bonds + π-bonds + lone pairs of electrons in the valence layer, i.e., the total number of valence electron pairs divided by 2. Step 3 Labelling the electron lone pairs between atoms For the CS2 molecule, the carbon atom (C) is less electronegative than the sulphur atom, so carbon is the central atom and sulphur is the outer atom. The central atom must be highly or minimally electronegative. Therefore, there are 4 and 6 valence electrons in the carbon and sulphur atoms respectively, so the total number of valence electrons in the CS2 molecule = valence electrons from 1 carbon atom + valence electrons from 2 sulphur atoms = 4 + 6(2) = 16. Step 1 Calculate the number of valence electrons for C and SĬarbon and sulphur are elements of groups 14 and 16 of the periodic table. Steps for drawing the CS2 Lewis structure Each sulphur atom (S) has two lone pairs of electrons. The carbon (C) and sulphur (S) atoms are each connected by a double bond. 
The CS2 Lewis structure consists of a central carbon atom (C) and two external sulphur atoms (S) bonded at 180°. 
III Marine Expeditionary Force (3 PAX)Ĥ.o. II Marine Expeditionary Force (3 PAX)Ĥ.n. Marine Corps Forces Central Command (2 PAX)Ĥ.l. Marine Corps Forces Command and North (2 PAX)Ĥ.k. Marine Corps Forces Pacific Command (2 PAX)Ĥ.j. Marine Corps Information Operations Command (1 PAX)Ĥ.i. Marine Corps Recruiting Command (2 PAX)Ĥ.h. 
Marine Corps Forces Cyber Command (1 PAX)Ĥ.g. Marine Corps Forces Reserve and South (2 PAX)Ĥ.f. Marine Corps Forces Europe and Africa (2 PAX)Ĥ.e. Deputy Commandant for Information (1 PAX)Ĥ.d. Deputy Commandant for Combat Development and Integration (1 PAX)Ĥ.c. Commanders will prioritize participation of COMMSTRAT directors, visual information officer and senior COMMSTRAT chiefs, to attend.Ĥ.a. It is strongly advised all Major Subordinate Commands and Major Subordinate Elements are included when selecting the appropriate personnel. The following commands are encouraged to send 45XX leadership resident within their commands to the OAG. The OAG is led by the COMMSTRAT MOS Sponsor and provides a critical forum to identify capability requirements and associated capability gaps across the doctrine, organization, training and education, materiel, leadership, personnel, facilities, and policy and cost (DOTMLPF-P/C)combat development pillars.Ĥ. The COMMSTRAT OAG is a forum for direct interface, planning and decision making between Headquarters Marine Corps, the operating forces and the supporting establishment concerning issues that affect the 4500 occupational field (OCCFLD). The OAG will be hosted by the COMMSTRAT MOS Sponsor – Director, Communication Directorate, Headquarters Marine Corps.ģ. POC/ARMISTEAD/MW/LTCOL/BRANCH HEAD/OCCFLDSPT/CD/TEL: (703) 692-1825/EMAIL: CHIEF/OCCFLDSPT/CD/TEL: (703) 614-1494/EMAIL: This calling message provides information for the COMMSTRAT OAG to be held 28 - 31 March 2023.Ģ. NARR/REFS A AND B PROVIDE AUTHORITY FOR THE 45XX COMMSTRAT OCCFLD MOS SPONSOR TO ESTABLISH AND HOST AN OAG. REF C PROVIDES FOR THE CONDUCT OF THE COMMSTRAT OAG. REF/A/MCO 5311.1E TOTAL FORCE STRUCTURE PROCESS// SUBJ/2023 COMMUNICATION STRATEGY AND OPERATIONS (COMMSTRAT) OPERATIONAL ADVISORY GROUP (OAG) CALLING MESSAGE//  
Hey Porter! also keeps prices low to ensure that they are accessible to design students from diverse backgrounds. Kahaza supports a variety of scripts including Arabic, Farsi, Urdu and Kurdish. 1 Display: Eclat, 2 Display: Bauhaus, 3 Display: Lobster. Display (decorative) type classification. They are typically suited for large point sizes and primarily used for display. So, if you’ve got a project that’s in need of a bold typography treatment, look no further than the authentic yet refreshing Kahaza. Display typefaces, also known as decorative, are a broad category of typefaces that do not fit into the preceding classifications. Tawfiq has also enlarged the dots (shaped more like diamonds) throughout the typeface which gives a “humanist appearance” while also giving the font a more “informal vibe” as Tawfiq puts it. To accentuate the modern feel, glyphs are elegantly expanded, but finished with distinct points, creating a feel that “blends the poise between sharpness and smoothness”. “The typeface takes a step beyond vintage influence by incorporating distinctive sharp characters that highlight detailed design work,” Tawfiq explains. 
This is where Hey Porter! really lives up to its experimental credentials, taking traditional aesthetics and graphic legacies and infusing them with touches that bring them firmly into the present day. It comes in regular, if you’re looking for a more clean and polished look, or bold for “a more impactful statement”.ĭespite being rooted in vintage aesthetics, Tawfiq was intent on ensuring that Kahaza still had a “dynamic contemporary balance”. These two influences make Kahaza a perfect display font, ideal for editorials, posters, branding, music videos and film titles, Tawfiq lists. (If you fancy a look at the rich legacy of material that has inspired Kahaza, we covered the Moving Pictures Painted book of cinema Egyptian posters from CentreCentre earlier this year.)Īlongside vintage film posters, Tawfiq also took inspiration for Kahaza from Naskh script, a form of lettering used commonly in newspaper and magazine headlines across the Arab world. It therefore comes as no surprise to hear that Tawfiq’s most recent typeface, Kahaza, is inspired by posters, specifically the titles you might find on vintage Egyptian film posters from the 1970s and 80s. To remedy his boredom while making infographics and reports through his work for an NGO, he created a poster every day for three years – often resulting in 5am bedtimes. When we first met Tawfiq Dawi, the man behind experimental type studio Hey Porter! back in 2021 we discovered that he had something of an affinity for posters. The capital letters H, I ,O and X all have both horizontal and vertical line symmetry.Īll the rest of the letters (F, G, J, K, L, N, P, Q, R, S and Z) have no line symmetry. It would be easy to draw a capital B that did have horizontal line symmetry-this one almost does. I can't tell that without actually flipping it upside down or measuring, though, so I left it in my list. Actually, even though I included B (and maybe you did too), B isn't perfectly symmetric in the Arial font because the top loop of the B is just a tiny bit smaller than the bottom loop. I see horizontal line symmetry in the captial letters B, C, D, E, H, I, O and X. Alphabets having both horizontal and vertical mirrors are H, I, O, X. I see vertical line symmetry in the capital letters A, H, I, M, O, T, U, V, W, X and Y (some of these have other kinds of symmetry too!) Alphabets having a reflection symmetry about a horizontal mirror are B, C, D, E, H, I, O, X. Even among san serif fonts, symmetry can be slightly different, so my answers may be different from yours if I write my letters differently. Arial is a san serif font-all of those extra bits are missing in an Arial font-that makes the letters have more symmetry. They are useful to have somewhere in the classroom so that when symmetry is discussed there are some examples that can easily be used. I'm using the Arial font, which is a san-serif font. Serifs are the little fancy bits on the ends of letters: Times is a serif font-f's have little lines at the bottom, and S's have little lines at the top and bottom, and A is thinner on the left side than the right side. These colourful and printable letters of the alphabet show the degrees of rotation and the reflective symmetry. It all depends on just how you draw your letters. This can be observed clearly from the figure given below. What letters of the English alphabet have reflectional symmetry i.e., symmetry related to mirror reflection about. In other words, it is defined as the sliding of an object about an axis. If the object is translated or moved from one position to another, the same orientation in the forward and backward motion is called translational symmetry. we get an equivalent equation.Introduction to Symmetry Line symmetry in capital letters syn- together + metron measure Reflection Symmetry The simplest symmetry is Reflection Symmetry (sometimes called Line Symmetry or Mirror Symmetry ). Reflection Symmetry Glide Symmetry Translation Symmetry. A circle with center at the origin and radius r >0 has equation x 2+ y 2= r 2. Show algebraically that a circle with center at the origin is symmetric with respect to the origin. If you end up with the exact opposite of what you started with (that is, if f (–x) = –f (x), so all of the signs are switched), then the function is odd.Īre circles symmetric with respect to the origin? To do this, you take the function and plug –x in for x, and then simplify. You may be asked to “determine algebraically” whether a function is even or odd. Alphabet Worksheets Printable Worksheets Geometrical Optics Learn Physics. In fact, all the letters of the English alphabet except G, Q, and V have reflection symmetry. symmetry Reflection symmetry in two directions This ray is not bent. 
It also makes components, such as gears, wheels, and axles, easy to make out of solid cubes or other polyhedra. If the resulting equation is equivalent to the original equation then the graph is symmetrical about the origin. This reflection symmetry is very important for creating looking artificial objects out of cubes or other polyhedra. Test for symmetry about the origin: Replace y with (-y) AND x with (-x). It is a standing line that divides the pattern into their replicating halves. If the line passes through the pattern from top to bottom, hence dividing the pattern into identical halves, then such line is the vertical line of symmetry. If you make the top smaller than the bottom, it has only one vertical line of symmetry. Some letters look the same when facing a mirror.Ĩ has two lines of symmetry and 180 degree rotational symmetry if you draw the top and bottom the same size. “H” and “I” have both vertical AND horizontal symmetry, as does “O” and “X.” But N, S, and Z don’t have reflectional symmetry at all! And they DO have rotational symmetry. O,T,U V,W,X, and Y.ĭoes the letter H have reflection symmetry? What letters of the English alphabet have reflectional symmetry (i.e., symmetry related to mirror reflection) about. 
What capital letters look the same in the mirror?Ĭapital English alphabet has 11 symmetric letters that appear same when looked at in a mirror. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
